How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
The heart is often one of the most underappreciated aspects of human anatomy, and its atrial appendages are often overlooked even in cardiac anatomy.
These delicate and astonishingly thin structures are challenges for medical device developers.
Some intricate structures within the heart are so thin that a business card can be read through them, said Tim Laske, VP of research and business development for the cardiac ablation solutions business at Medtronic. When observing the heart in a surgical space, it becomes evident that atrial appendages carry blood, emphasizing the need to visualize their unique properties and thickness.
Rather than relying solely on “textbook depictions,” designing medical tubing that accommodates the true characteristics of this delicate anatomy is crucial for optimal device performance.
“The ventricular anatomy is incredibly complex,” Laske said during a presentation at DeviceTalks Boston. “Everyone has a slightly different anatomy, and their conduction system is slightly different. Please consider that when designing devices.”
By acknowledging these differences, engineers can develop tubing solutions that seamlessly integrate with the diverse ventricular anatomy, ultimately enhancing device performance and patient outcomes.
Nitinol: Unveiling the characteristics of an alloy
Composed of an atomic mixture of nickel and titanium, nitinol possesses distinct properties that make it a preferred choice for medical tubing. In the context of tubing design, two particularly interesting properties come to light.
First, nitinol has excellent thermal connectivity, boasting efficient conductivity that proves invaluable when used as an electrode. This property ensures optimal heat dissipation, enhancing the performance of devices utilizing nitinol tubing as an electrode component.
Secondly, nitinol demonstrates exceptional elasticity, making it an ideal material for applications that require high flexibility without the risk of kinking. Nitinol tubing can endure significant deformation without experiencing plastic deformation, making it particularly well-suited for guidewires and other critical areas where flexibility and shape retention are paramount.
“It’s not only more compliant and flexible than stainless steel but it can be deformed to a much greater extent without plastic deformation. Hence, it’s used in guidewires and other areas where you need something highly flexible that won’t kink,” Laske said.
Nitinol’s unique properties extend beyond its thermal connectivity and elasticity. Two notable phenotypes associated with nitinol are shape memory and super elasticity.
Shape memory allows nitinol to be heat-treated into a specific shape and then deformed. However, nitinol returns to its original shape upon reheating past its transition temperature. This characteristic enables the customization and adaptation of tubing to suit various anatomical requirements, leading to enhanced patient comfort and procedural success.
Superelasticity, on the other hand, allows nitinol to withstand substantial strain and stress while retaining its original shape. This property is particularly advantageous in medical devices that undergo significant mechanical loads, ensuring longevity, reliability and safety.
“This shape change is important if you’re using it for the shape memory phenomenon,” Laske said.
Medical tubing made from nitinol offers several advantages. Its biocompatibility and biostability make it suitable for introduction into the body, while its flexibility minimizes trauma and reduces the risk of damage. Moreover, nitinol’s ability to undergo substantial size changes, from a small initial form to a larger expanded state within the body, enhances its versatility and applicability in various medical procedures, most notably less-invasive catheter delivery of cardiovascular implants.
How to process nitinol for medical tubing
Processing nitinol typically involves drawing it into wire for applications like guidewires or embolic coils. However, laser cutting from tubing is the more common approach.
Other processes such as electropolishing ensure a smooth surface finish. Smoothness is crucial in preventing the formation of titanium nitride inclusions, which can lead to fractures.
When selecting a nitinol vendor, it is essential to work with reputable sources and inspect the material for any inclusions that may compromise its performance, Laske said.
“Anyone that’s worked with nitinol, you know your enemy is titanium nitride inclusions. You can get titanium nitride crystals, which can be pretty large, and those will nucleate fractures. So when you’re choosing your nitinol vendor, make sure you’ve got a very reputable house and then when you’re inspecting it, look for these inclusions and be sure that they don’t exist if it’s in a high-fatigue-state environment.”
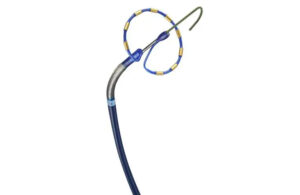
Applications of nitinol-based tubing
One of the more widely known applications of nitinol is in transcatheter heart valves, which are designed to replace heart valves non-surgically, Laske said. In these procedures, arteries and veins are used as “superhighways” to access the heart. When using a nitinol stent in this application, engineers should consider the durability once placed in the heart and how small the device can be compressed for minimal impact on the vasculature. It also has to crimp down inside a sheath to access the heart and then spring into place at the proper location in the heart.
Medtronic uses nitinol in a range of devices.
Medtronic uses self-expanding nitinol for the frame of its Harmony transcatheter pulmonary value, with nitinol wires hand-sewn together in zigzags.
Nitinol also allowed Medtronic’s Micra leadless pacemaker to be placed in the heart.
“It had a battery life of about six months, which was not enough to place inside the heart,” Laske said. “As battery technology evolved and with clever use of nitinol, the device can now be placed inside the heart as a permanent pacemaker.”
Micra has FlexFix nitinol tines that come out of the device’s sheath in the body to engage with the tissue and secure the device in place. If a physician needs to recapture the device, it can still be pulled back with the nitinol tines in place.
Medtronic also uses nitinol in its Arctic Front Advance cryoablation platform, Laske said. That system uses nitrous oxide to freeze the pulmonary veins to kill the diseased tissue circumferentially around the cardiac veins.

“This wouldn’t seem like it’s an obvious place to use nitinol,” Laske said. “But there are two different components.”
First, the Achieve Advance mapping catheter must navigate the pulmonary veins. Laske said the device needs to be able to travel in a straight line to the veins, then deploy and be manipulated while still able to return to its original shape.
The injection tube that the nitrous oxide runs through is also made of nitinol and can adapt to the shape of the pulmonary vein. Liquid nitrous oxide is then pumped down the nitinol tube and injected at the proper site.
Medtronic’s Affera and PulseSelect mapping and ablation systems use nitinol in their components as well. The Affera Sphere 9 catheter has a nitinol mesh that is super elastic so that it can be compressed into a small catheter and expand within the body.
“[The device] needs to come out of a small catheter and go into a large configuration. It has good electrical conductivity, so it can be used as an electrode. It has well-understood biocompatibility and we know how it behaves inside the body into the bloodstream,” Laske said.
Article Source:MTE









