Tractus Vascular Marks First Human Use of its Support Catheter
The Tractus Crossing Support Catheter is FDA 510(k)-cleared with 0.014-inch, 0.018-inch, and 0.035-inch guidewire compatibility……
Read More
Double-Digit Growth Forecast for 3D Printed Medical Devices Market
Market value to more than triple as collaborations and improved surgical success rates boost demand…….

Why You Should Embrace Direct-to-Consumer Lab Testing
Of its many wiles, one of the biggest impacts that the pandemic has had on society is that it forced much of the world into an existential crisis. Ever since COVID-19 made its debut, people from across the globe have been reflecting on their lives, careers, and health in profound, soul-searching ways……
Read More
Penta Hospitals Partners with Siemens Healthineers to Enhance Healthcare in Slovakia
The strategic 15-year Value Partnership will significantly improve patient care provided by the flagship Bory Hospital starting in 2023……

Discovering the Value of Surface Treatments and Coatings for Medtech
Surface treatments and coatings enable device makers to design more benefits into their medical technologies……

Medical Device Servicing vs. Remanufacturing
Each are distinct processes, but both rely on quality management……

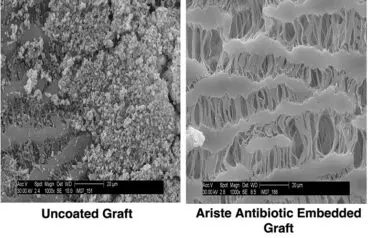
Ariste Medical co-founder sees great potential for drug-coated implants and orthopedics
You can expect more to come from that technology, Jennings told Medical Design & Outsourcing in an interview covering future applications, the drug-device development process and what she’s learned through it all……
Medtec China 2022 is now open for visitor registration, helping medical device manufacturers in Medtech sourcing and Supply Chain stabilization
How to stabilize supply chain nowadays has become one of the most important topics to most medical device manufacturers all the world. As an international high-quality one-stop sourcing platform for the upstream of China’s medical device manufacturing……

The Problem with Medical Plastics, and How to Fix It
The ubiquity of plastics in medical applications has tremendously benefited healthcare, but it also bears an environmental cost……
Assessment of Medical Devices Without Animal Experiments
To ensure patient safety, regulations such as the EU Medical Device Regulation require risk assessments to be conducted for all medical devices, including biological evaluation in accordance with ISO 10993. This article explores how manufacturers can have their products assessed efficiently without animal experiments……
