How Are Hypodermic Needles Made?
It’s a marvelous invention, but most don’t think much about it unless they work in healthcare……

Silicone Tech Advances are Key for In Vitro Device Manufacturers
Advances in silicone technology help in vitro device manufacturers respond to the demands of an accelerating market……

The Advantages of Bioburden Screening for Sterilization Validation
Bioburden can be introduced at several stages throughout the manufacturing process……

AI, ML, & Cybersecurity: Here’s What FDA May Soon Be Asking
FDA has released a number of documents that could help clarify its expectations for artificial intelligence, machine learning, and cybersecurity……
Read More
New 3D Printing Technique: A Game Changer for Medical Testing Devices
The technique fabricates microfluidics for biomedical applications at a microscale not previously possible……

Letting the Light In: How Failure Analysis is the Cornerstone of Success
Failure analysis is a vital part of the process of developing medical devices that function safely and efficiently……

Prototyping with DIN Rail
DIN rail assemblies can be a great option for one-off prototypes where changing requirements are expected……

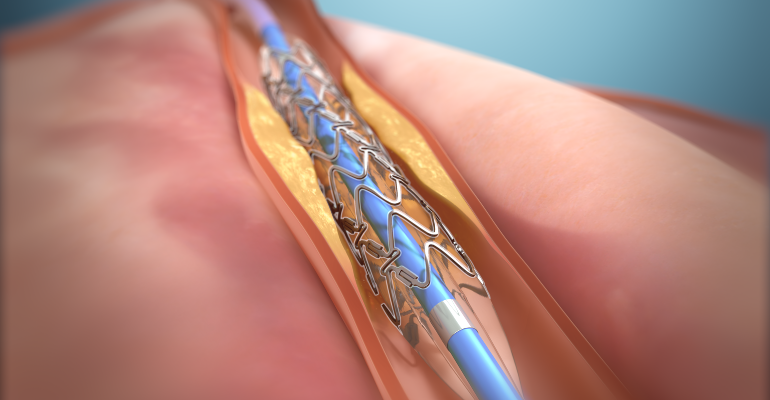
New Flexible Coronary Artery Implant Enables Vessel Motion
The DynamX Bioadaptor is designed to restore vessel motion and function……
An Update on Surgical Robotics
Intuitive is still the 800-pound gorilla in the room, but the entire market segment struggles to prove its value and justify its high cost……

A Haystack with No Needle: Is an Extractables Study Appropriate for Limited-Contact Devices?
Based on ISO 10993-18, some medical device manufacturers to wonder if an extractables study is required for all devices, regardless of duration of contact. Experts at Nelson Labs share their knowledge on the matter……
